How Do You Know if Water Is Produced in a Chemical Reaction
6.4 Chemical equations and chemical reactions
The previous section shows how dissimilar elements tin can either exist on their own or combine with other elements to make compounds. This section builds on these ideas past looking at chemical reactions in more particular. It besides shows how chemical shorthand can be extended to describing chemical reactions.
First, consider some of the molecules described earlier: water, methane, carbon dioxide and ammonia.
Question 26
What are the formulas for each of these four molecules?
Answer
The formulas are H2O, CHfour, CO2 and NH3, respectively.
Although you don't need to retrieve the chemic formulas of compounds introduced in this class, you'll probably find it useful to memorise a few such every bit those for the 4 molecules above, plus those for hydrogen (H2), nitrogen (Due north2) and oxygen (Oii) molecules. This volition help you lot in reading and writing these chemic formulas without continually referring dorsum to previous sections.
Now you lot will look at reactions involving the elements hydrogen, carbon and oxygen and the compounds methane, h2o and carbon dioxide.
Hydrogen will react with oxygen when it is ignited (it is quite explosive) to class water. To write such a reaction in terms of a chemical equation, the substances that undergo the reaction are put on the left and the substances that are produced in the reaction are put on the right. The reactants on the left are linked to the products on the right past an arrow.
The equation tin can be written every bit a discussion equation 'hydrogen and oxygen brand water' or, using a little chemical shorthand, it tin can be written every bit:
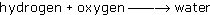
where the arrow means 'goes to'.
At present, endeavor using chemical autograph to write the equation. Substituting symbols in the word equation gives:
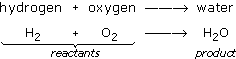
The equation shows the reactants on the left of the arrow being converted to the product on the right. However, there is something wrong with this equation. You can see what is wrong by looking at Figure 21 where the reactants and products are shown equally a diagram.
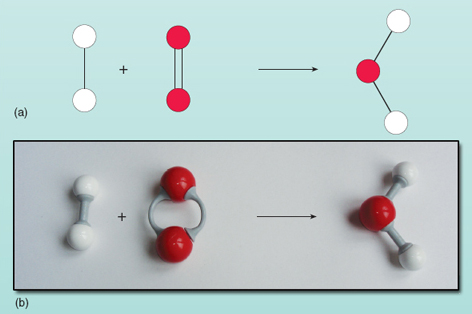
Effigy 21 The unbalanced equation for the reaction betwixt hydrogen and oxygen to requite water: (a) diagrammatic representation; (b) ball-and-stick model. Note that the oxygen molecule comprises two oxygen atoms linked by a double bond. This is because each oxygen atom has two 'hooks' to link up
Counting the numbers of atoms on each side of the equation shows that there are two oxygen atoms on the left compared with one on the correct. The number of oxygen atoms on both sides of the equation must be equal - they can't magically appear and disappear during a reaction. Information technology is not possible to change the limerick of the water molecule on the right of the equation as the water molecule exists as a grouping of two hydrogen atoms and 1 oxygen atom all bonded together. Each oxygen molecule supplies two oxygen atoms so will always form 2 h2o molecules, provided there are 2 hydrogen molecules (each existence a unit of two atoms) to react with it. Thus two molecules of hydrogen and one molecule of oxygen are needed to brand ii molecules of water. The reaction is accurately expressed by:

There are now the same numbers of each type of atom on both sides of the equation: the chemical equation is balanced, so nosotros can now supersede the arrow with an equals sign.
A terminal tidy-upwardly to avoid repeating the hydrogen molecules on the left of the equation and the water molecules on the right is to correspond them by 2H2 rather than Htwo + H2. So the counterbalanced chemic equation becomes:
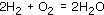
This is shown diagrammatically in Figure 22.
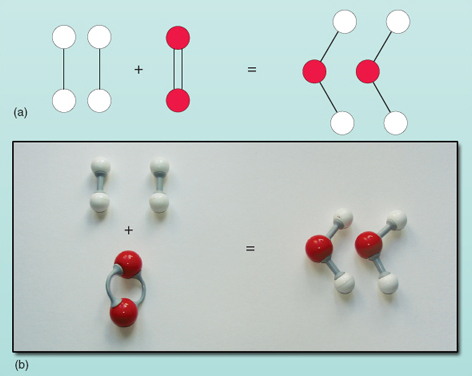
Figure 22 The balanced equation for the reaction between hydrogen and oxygen to give water: (a) diagrammatic representation; (b) ball-and-stick model
Chemical equations evidence in a very concise way not only which atoms and molecules react together to form the products but likewise how many of each sort of atom and molecule are involved. It is important to think that the number before a molecule means the number of that particular molecule. For example 3HtwoO means 3 molecules of water, giving a full of half dozen hydrogen and three oxygen atoms.
Chemic equations must balance; the number of atoms of each type of element on both sides of the equation must exist equal.
You at present know how to 'read' or interpret a chemical equation - and how it represents a chemical process. However, chemists are also very adept at writing chemical equations every bit a form of shorthand to describe various chemical processes or reactions. Writing chemical equations is like writing in whatsoever foreign linguistic communication - it requires practice to become fluent. Nevertheless, all budding scientists accept to start somewhere and the section below shows you how to practice this using a very familiar chemical reaction. If you wish to check whether you demand to written report this section, try Question 20 first.
Source: https://www.open.edu/openlearn/nature-environment/the-environment/environmental-studies/water-life/content-section-6.4
Postar um comentário for "How Do You Know if Water Is Produced in a Chemical Reaction"